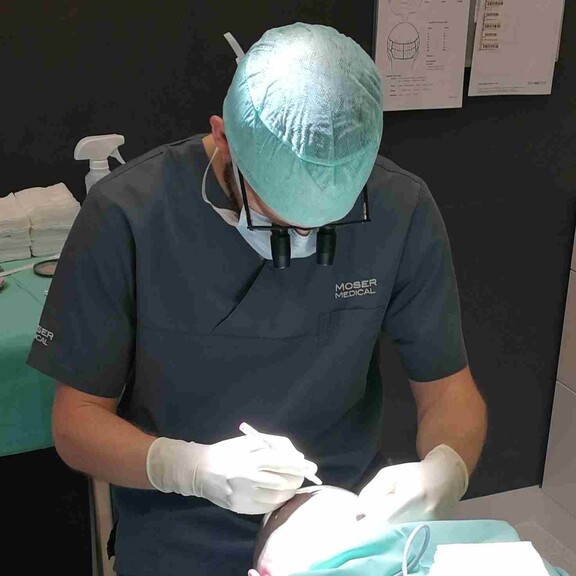
Hair Transplantation at Moser Medical
A full head of hair - thanks to Austria’s pioneers in beautiful hair
Decades ago, international hair researchers discovered that the hair on the back of the head is genetically programmed to grow for a lifetime. This discovery forms the basis effective hair loss solutions – hair transplants at Moser Medical.
Moser Medical is a pioneer in the transplantation of a patient’s own hair and number one in Europe. Since 1979, our specialists have conducted research and performed procedures in this area. It is important to know that hair roots naturally grow in groups of 1, 2, 3 or 4 hairs. These groups are called follicular units (FUs) or grafts. FUs from the back of the head are programmed to grow throughout our lives. They are therefore taken and transplanted onto bald or thinning areas of the scalp where they continue to grow and form new hair.
The best medicine for hair loss is your own hair.
Moser Medical facts & figures
- Experience of more than 35,000 procedures
- Strip and follicular unit extraction methods (FUT & FUE)
- State-licensed clinic
- 17-person medical team
- Innovative nutrient solution (“storage solution”) from our own R&D
- Moser Medical BioRegeneration
- Continuous further training and development
- International awards
- Our own research lab
- The highest hygiene standards
- Intensive pre & post-care
The quickest, easiest way to full hair
If I had known how easy and uncomplicated a hair transplant is, I would have gone to Moser Medical years ago. The procedure has definitely brought greater quality to my life!
Criteria for transplant using your own hair
At Moser Medical, you can choose the extraction method
Individual extraction (FUE technique)
Hair follicles are removed individually from the donor area with the follicular unit extraction method. Moser Medical is the first provider in Austria to use the innovative hybrid rotation oscillation instrument (“HyRO”), which enables a particularly gentle treatment of the highest quality. This removal technique guarantees the highest possible growth rates, but requires shaving the hair on the back of the head to 1 mm in length.
NEW: Long hair FUE method. This method allows hair to be extracted hair without shaving. The advantage is clear: there are barely any visible signs of treatment, and you can get back to your everyday life quickly.
Strip extraction (FUT technique)
Used for decades, the follicular unit transplantation method involves extracting the donor hair in an elliptical strip of hair from the back of the head. From this strip, the individual follicles are extracted precisely under microscopes. FUT is also known as the strip extraction method. The procedure for implanting the hair is identical for both FUT and FUE extraction methods.
Hair transplantation for men and women
Our transplants are naturally open to all patients, regardless of gender. Please keep in mind the different criteria that need to be considered when choosing the most suitable treatment method.
Signs of quality at Moser Medical
We only perform procedures where all criteria for a successful hair transplant with the highest quality and long-term security have been carefully considered. Moser Medical patients benefit from our decades of experience, based on the results of tens of thousands of successful hair transplants.
Our experienced team of specialists and medical assistants perform the highest-quality hair transplants, and they are passionate about achieving the best results with every procedure. All hair transplants worldwide are measured using this “gold standard.”
As a member of the ISHRS (International Society of Hair Restoration Surgery), the largest and most active international organization of its kind, Moser Medical is among the leading hair transplant practitioners in the world. Founded in 1992, the ISHRS non-profit initiates and promotes the continuous improvement of quality, as well as the training of hair restoration surgery professionals.
Moser Medical is not only a registered member of the ISHRS, but also participates in conferences to exchange information and experiences with other leading specialists from around the globe. Our specialists frequently give lectures at hair transplant conferences and use them as an opportunity for further training and development.
Together we discuss which extraction method is best suited to your current situation and the results you want during your personal consultation.
Your advantages with a hair transplant
- Experience: Hair transplantation specialist since 1979.
- The most modern techniques: Only at Moser Medical can you decide between the most important methods (FUT, FUE and long hair FUE).
- Quality: We are always one step ahead, we don’t follow every trend, and know how our patients will look in 10, 20 or more years, thanks to our experience.
- Safety: Our unique expertise and experience enables best results and gives you highest safety regarding the expected results.
- Maximum performance: Internationally recognized hair surgeons and an 17-person experienced medical assistant team.
- Recognized: Awarded the “Oscar of hair restoration surgery” (Platinum Follicle Award) and many other international distinctions.
- The best hygiene: Our state-licensed private clinic in Vienna, Austria, exceeds the highest European standards and the AKH standard.

Problem areas and the right solution
A hair transplant can offer the solution for many cosmetic problems, and can be flexible depending on your degree of hair loss, hair appearance and the location of the problem. The following issues and problem areas can be addressed with hair transplantation at Moser Medical, for both women and men:
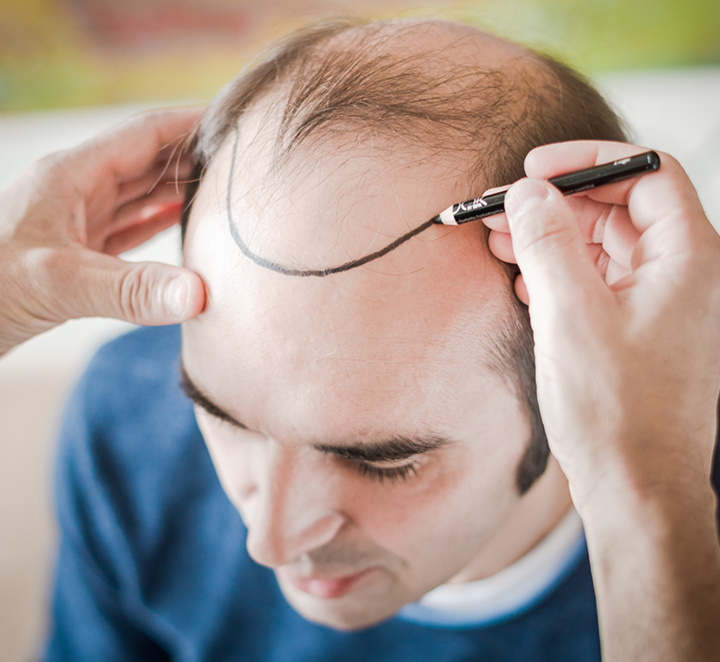
Hair transplant consultation
Are you interested in a hair transplant by Moser Medical in Vienna? Our experienced team are happy to discuss your options.












Hair transplant consultation – not only in Vienna
Moser Medical offers a free, non-binding initial consultation not only in Vienna, but across Austria. We will explain possible procedures and their prospects of success, as well as the entire procedure. Contact us today to schedule your consultation at one of our locations in Austria!